-
Clinical trials begin for new Ebola vaccine developed by University of Oxford
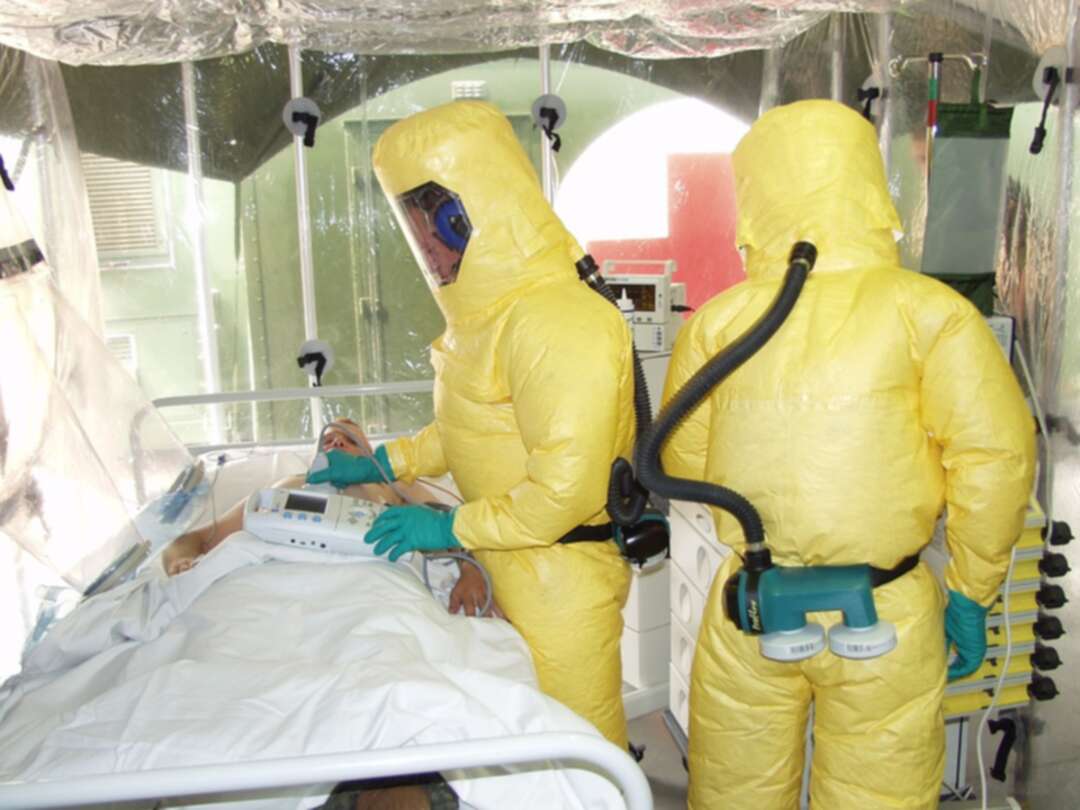
The BBC reported that clinical trials have begun for a new Ebola vaccine developed by the University of Oxford.
It said that the jab has been designed to tackle the Zaire and Sudan types of Ebola, which together have caused nearly all Ebola outbreaks and deaths worldwide.
It mentioned that the University of UKhas launched phase one of its trials, testing the vaccine in human volunteers.
The BBC added, Ebola vaccines exist for the Zaire species but Oxford researchers hope the new jab will have a wider reach.

Teresa Lambe, lead scientific investigator at the University of Oxford, said: "Sporadic Ebolavirus outbreaks still occur in affected countries, putting the lives of individuals, especially frontline health workers, at risk. We need more vaccines to tackle this devastating disease."
Read more: South Africa’s former president FW de Klerk dies at 85
There are four species of Ebola virus that have been known to cause disease in humans. Of these, Zaire is the most lethal, causing death in 70% to 90% of cases if left untreated.
The new vaccine developed by Oxford scientists is based on a weakened version of a common cold virus that has been genetically modified so that it is impossible for it to replicate in humans.
Read more: Virus responsible for Covid-19 detected in a pet dog in the UK
This method has already been used successfully in the Oxford-AstraZeneca Covid-19 vaccine.
Phase one of the trials will see 26 people aged 18 to 55 receive one dose of the ChAdOx1 biEBOV Ebola vaccine at the university. They will then be monitored over a six-month period, with results expected in the second quarter of 2022.
Source: BBC
You May Also Like
Popular Posts
Caricature
BENEFIT Sponsors BuildHer...
- April 23, 2025
BENEFIT, the Kingdom’s innovator and leading company in Fintech and electronic financial transactions service, has sponsored the BuildHer CityHack 2025 Hackathon, a two-day event spearheaded by the College of Engineering and Technology at the Royal University for Women (RUW).
Aimed at secondary school students, the event brought together a distinguished group of academic professionals and technology experts to mentor and inspire young participants.
More than 100 high school students from across the Kingdom of Bahrain took part in the hackathon, which featured an intensive programme of training workshops and hands-on sessions. These activities were tailored to enhance participants’ critical thinking, collaborative problem-solving, and team-building capabilities, while also encouraging the development of practical and sustainable solutions to contemporary challenges using modern technological tools.
BENEFIT’s Chief Executive Mr. Abdulwahed AlJanahi, commented: “Our support for this educational hackathon reflects our long-term strategic vision to nurture the talents of emerging national youth and empower the next generation of accomplished female leaders in technology. By fostering creativity and innovation, we aim to contribute meaningfully to Bahrain’s comprehensive development goals and align with the aspirations outlined in the Kingdom’s Vision 2030—an ambition in which BENEFIT plays a central role.”
Professor Riyadh Yousif Hamzah, President of the Royal University for Women, commented: “This initiative reflects our commitment to advancing women in STEM fields. We're cultivating a generation of creative, solution-driven female leaders who will drive national development. Our partnership with BENEFIT exemplifies the powerful synergy between academia and private sector in supporting educational innovation.”
Hanan Abdulla Hasan, Senior Manager, PR & Communication at BENEFIT, said: “We are honoured to collaborate with RUW in supporting this remarkable technology-focused event. It highlights our commitment to social responsibility, and our ongoing efforts to enhance the digital and innovation capabilities of young Bahraini women and foster their ability to harness technological tools in the service of a smarter, more sustainable future.”
For his part, Dr. Humam ElAgha, Acting Dean of the College of Engineering and Technology at the University, said: “BuildHer CityHack 2025 embodies our hands-on approach to education. By tackling real-world problems through creative thinking and sustainable solutions, we're preparing women to thrive in the knowledge economy – a cornerstone of the University's vision.”
opinion
Report
ads
Newsletter
Subscribe to our mailing list to get the new updates!